Why Nutrients Matter More Than You Think in Hydroponics
By Hydro Experts
In soil gardening, the growing medium itself supplies a buffer. Organic matter breaks down, microbial activity converts compounds, and the soil chemistry moderates what roots actually receive. In hydroponics, none of that exists. The nutrient solution is the entire feed source. What you put in the reservoir is what the plant gets, in the concentration and ratio you provide. There is no correction mechanism between the solution and the root.
This is why hydroponic nutrients explained properly goes beyond listing elements on a bottle label. The ratios matter, the timing matters, the form of each element matters, and the interactions between elements matter. A grower who understands these variables produces healthier plants and higher yields. A grower who does not tends to encounter deficiencies, toxicities, and poor results that look like bad luck but trace back to nutrition.
The Basic Framework: What Plants Need and Why
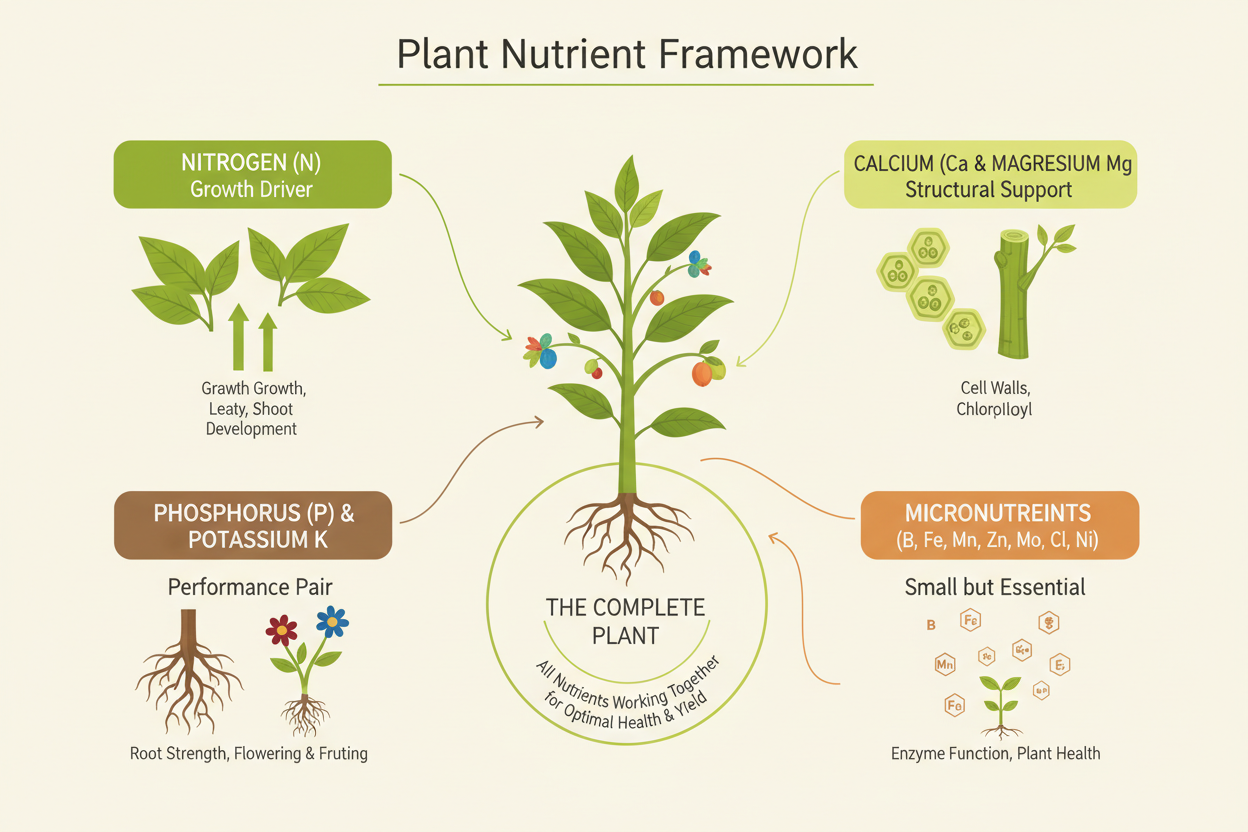
Plants require seventeen essential elements to complete their life cycle. Carbon, hydrogen, and oxygen come from water and air. The remaining fourteen come from the nutrient solution in hydroponics.
These fourteen divide into macronutrients and micronutrients, a distinction based on the quantity required rather than importance. A micronutrient deficiency is just as capable of stopping plant growth as a macronutrient deficiency.
Macronutrients divide further into primary and secondary categories.
- Primary macronutrients are nitrogen (N), phosphorus (P), and potassium (K). These appear on every nutrient label and are required in the largest quantities across all growth stages.
- Secondary macronutrients are calcium (Ca), magnesium (Mg), and sulphur (S). Plants need these in substantial quantities, and deficiencies are common in hydroponic systems that use soft water or poorly formulated base nutrients.
- Micronutrients include iron (Fe), manganese (Mn), zinc (Zn), copper (Cu), boron (B), molybdenum (Mo), chlorine (Cl), and nickel (Ni). Required in trace amounts, but each one plays a specific biochemical role that cannot be substituted.
Nitrogen: The Growth Driver
Nitrogen is the element most directly tied to vegetative growth. It is a core component of amino acids, proteins, chlorophyll, and nucleic acids. Without adequate nitrogen, plants produce less chlorophyll, leaves yellow from older growth upward, and vegetative development slows or stops.
In hydroponics, nitrogen is supplied in two forms: nitrate (NO3-) and ammonium (NH4+). The ratio between these forms affects root zone pH, uptake rate, and how the plant allocates the nitrogen it receives. Nitrate is the preferred form for most crops in recirculating systems because it is stable, has a predictable uptake rate, and does not acidify the root zone as aggressively as ammonium.
High nitrogen through the flowering or fruiting stage suppresses reproductive development. This is a common grower error: continuing a vegetative feed formula into flowering, resulting in lush foliage and poor fruit set. Nitrogen requirements drop as the plant transitions, and a nutrient mix for hydroponics designed for the full growth cycle accounts for this shift.
Phosphorus and Potassium: The Performance Pair
Phosphorus drives energy transfer within the plant. It is central to ATP production, root development, flowering initiation, and seed formation. Phosphorus deficiency manifests as purpling of leaf undersides and stems, particularly in younger plants, and reduced root mass.
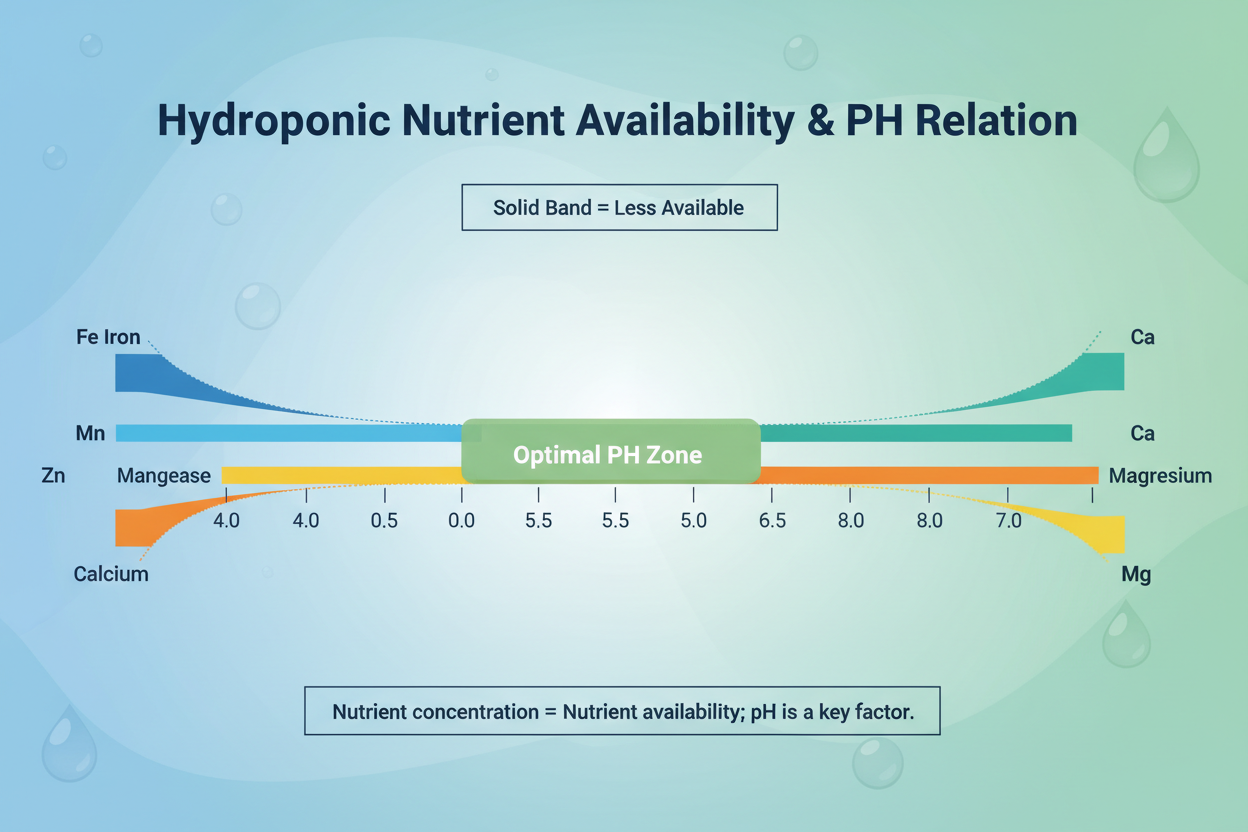
In hydroponics, phosphorus availability is highly pH-dependent. At pH above 7.0, phosphorus precipitates and becomes unavailable even if the concentration in solution appears adequate. Maintaining solution pH in the 5.5 to 6.5 range keeps phosphorus soluble and accessible.
Potassium supports a wider range of plant functions than its reputation as a "flowering nutrient" suggests. It regulates stomatal opening and closing, controls water uptake, activates enzymes, and supports phloem loading of sugars. During fruiting, potassium demand increases substantially. A solution that does not increase potassium supply relative to nitrogen during this stage limits fruit quality and size regardless of other inputs.
The N:P:K ratio on a nutrient label expresses the relative proportion of each element. A vegetative formula might carry a ratio weighted toward nitrogen (e.g., 3:1:2). A bloom formula shifts toward phosphorus and potassium (e.g., 1:3:2). Understanding what the ratio means and when to apply which formula is foundational to productive hydroponic nutrition.
Calcium and Magnesium: Overlooked Until Something Goes Wrong
Calcium is a structural element. It forms calcium pectate, which binds cell walls together and gives plant tissue its rigidity. It also plays a signalling role in stress responses. Calcium deficiency causes tip burn in leafy crops, blossom end rot in tomatoes and capsicums, and distorted new growth, because calcium moves through the plant only via the transpiration stream and does not redistribute from older tissue.
Magnesium sits at the centre of every chlorophyll molecule. It is also required for phosphorus uptake and enzyme activation. Magnesium deficiency appears as interveinal chlorosis on older leaves: the veins stay green while the tissue between them yellows. Growers often mistake this for an iron deficiency and add iron without effect, because the problem is magnesium.
Both calcium and magnesium are frequently undersupplied in hydroponic systems where the base water is very soft (low in dissolved minerals) or where the nutrient formulation does not account for them explicitly. A dedicated Cal-Mag supplement resolves this in most systems and is a standard addition to any serious nutrient program.
Micronutrients: Small Quantities, Critical Functions
Iron is the micronutrient most commonly deficient in hydroponic systems. It is required for chlorophyll synthesis and electron transport. Deficiency appears as yellowing of new growth while older leaves remain green, the reverse pattern from magnesium deficiency. Iron availability collapses above pH 6.5 in hydroponic solution, which is why pH management and iron supply are closely linked.
Manganese works alongside iron in photosynthesis and enzyme activation. Zinc is required for auxin synthesis, which controls cell elongation and root development. Copper is involved in photosynthesis and protein metabolism. Boron supports cell wall formation, pollen viability, and sugar transport. Molybdenum is required for nitrogen metabolism, particularly for converting nitrate to ammonium within the plant.
Each of these elements is required in quantities measured in parts per million or less. The margin between deficiency and toxicity for some micronutrients, particularly copper and boron, is narrow. A well-formulated hydroponic nutrient kit includes all micronutrients in chelated form at appropriate concentrations, removing the need for the grower to source and measure individual trace elements.
Chelation refers to the process of binding a metal ion to an organic molecule that keeps it soluble across a wider pH range. Chelated iron, for instance, remains available at pH levels where unchelated iron would precipitate. Quality nutrient formulations use chelated micronutrients as standard.
Nutrient Ratios and Growth Stage Matching

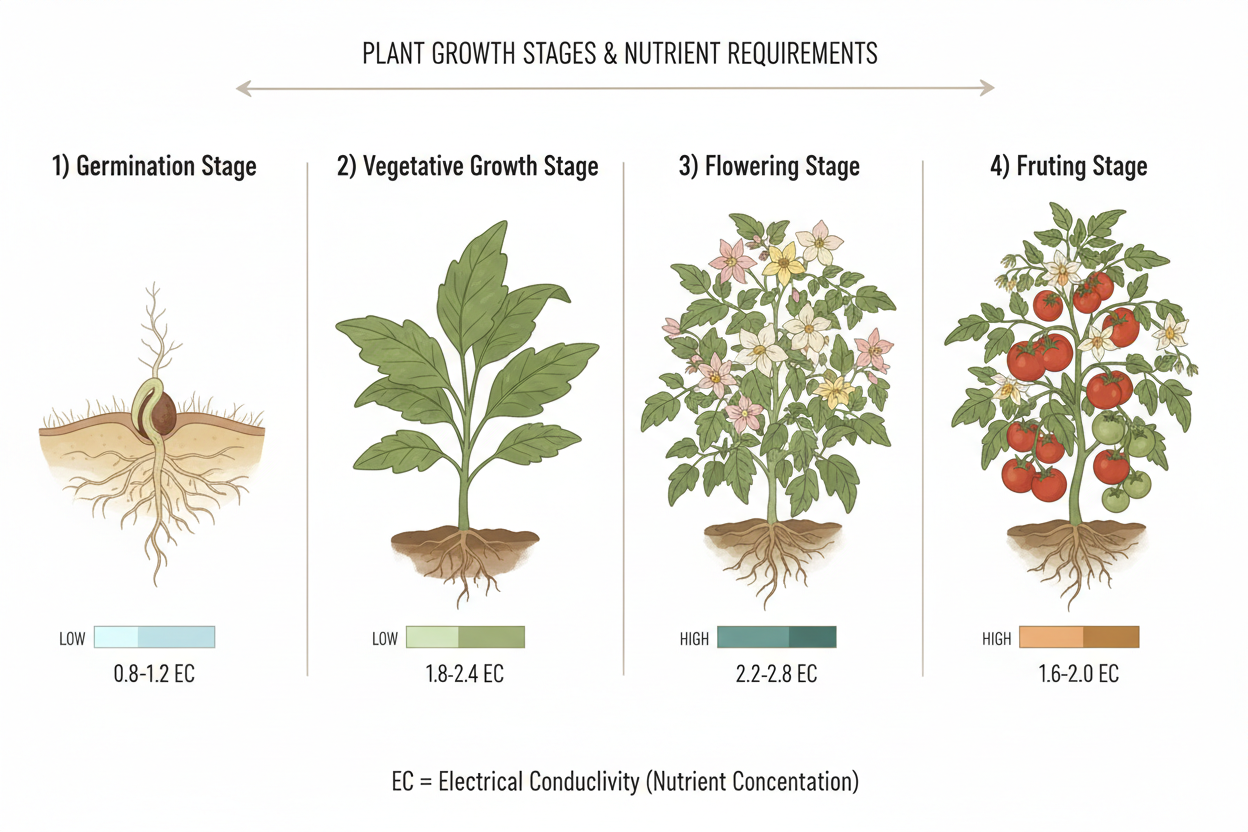
The nutrient requirements of a plant are not static. They shift across germination, vegetative growth, flowering, and fruiting. A single-formula approach applied across the full growth cycle produces adequate results at best and deficient or excessive nutrition at worst depending on the stage.
During germination and early seedling development, nutrient concentrations should be low. Young roots are sensitive and easily burned by high EC (electrical conductivity, the measure of dissolved nutrient concentration in solution). A starter solution at 0.8 to 1.2 EC suits most seedlings.
Vegetative growth demands higher nitrogen, moderate phosphorus, and moderate potassium. EC can increase to 1.6 to 2.4 depending on the crop and system. Calcium and magnesium requirements increase as leaf area expands.
At the transition to flowering, nitrogen reduces relative to phosphorus and potassium. Some growers run a transitional flush or reduce feeding concentration briefly to encourage the plant to shift its energy allocation from leaf to flower production. EC during flowering typically holds between 1.8 and 2.6 for most crops.
Late fruiting stage sees continued high potassium demand. Some formulations introduce a specific ripening or late-stage nutrient designed to support fruit density and flavour development. Pre-harvest flushing with plain water or a flushing agent for one to two weeks is standard practice to clear residual nutrient accumulation from plant tissue.
Matching the nutrient mix to the growth stage is the single most controllable variable in hydroponic nutrition management.
pH and EC: The Two Numbers That Govern Everything
Nutrient concentration and nutrient availability are different things. A solution can contain adequate quantities of every element and still deliver deficient nutrition if pH is out of range.
The optimal pH window for most hydroponic crops is 5.5 to 6.5. Within this range, all essential elements remain soluble and available. Outside it, specific elements drop out of availability in predictable patterns. Iron, manganese, and zinc become unavailable as pH rises above 6.5. Calcium and magnesium availability drops below pH 5.5.
EC measures total dissolved solids in solution but does not tell you what those solids are. A high EC from nutrient accumulation in a recirculating system can coexist with a specific element deficiency if that element has been preferentially taken up by plants and not replaced. Top-feeding reservoirs with fresh solution and running drain-to-waste systems are methods of managing nutrient drift over time.
Check pH and EC daily in active systems. Adjust pH using food-grade phosphoric acid (pH down) or potassium hydroxide (pH up). Record readings over time to identify trends. A pH that drops consistently indicates biological activity in the reservoir or acid-forming nutrient chemistry. A pH that rises consistently suggests the opposite or that plants are preferentially taking up anions.
Hydro Experts: The frontrunner for Australian Hydroponics
Hydro Experts Australia supplies nutrient kits and various other products formulated for the full growth cycle. The kits remove the complexity of sourcing and mixing individual components and are calibrated for use in Australian water conditions, which vary significantly between regions and affect how base nutrients perform. Visit our website now!
FAQs
Macronutrients are required in larger quantities and include nitrogen, phosphorus, potassium, calcium, magnesium, and sulphur. Micronutrients are required in trace amounts and include iron, manganese, zinc, copper, boron, molybdenum, chlorine, and nickel. Both categories are essential. A deficiency in any micronutrient is as capable of halting plant development as a macronutrient deficiency, despite the smaller quantities involved.
In recirculating systems, top up the reservoir with fresh solution daily to replace water taken up by plants, and perform a full reservoir change every one to two weeks. As plants take up nutrients selectively, the ratio of elements in the solution drifts from the original formulation. A full change resets this. In drain-to-waste systems, fresh solution is used each irrigation cycle, so drift is not a concern.
The most common cause is pH outside the 5.5 to 6.5 range. Elements are present in solution but unavailable to roots because they have precipitated or their uptake is chemically blocked. Check and correct pH first before adjusting nutrient concentration. The second most common cause is using a formula designed for one growth stage during a different stage, resulting in an imbalanced ratio for the plant's current requirements.
Soil nutrients are formulated for a different uptake environment. Many contain organic compounds designed to be broken down by soil microbes before plant uptake, which does not occur in hydroponic systems. Some components can block drip emitters, foul reservoirs, or create pH instability. Use nutrients specifically formulated for hydroponics to ensure solubility, correct elemental ratios, and compatibility with the system.
EC (electrical conductivity) measures the total concentration of dissolved salts in nutrient solution, expressed in millisiemens per centimetre (mS/cm). It indicates nutrient strength but not nutrient composition. Target ranges vary by crop and growth stage: seedlings perform well at 0.8 to 1.2 mS/cm, vegetative growth at 1.6 to 2.4 mS/cm, and flowering and fruiting at 1.8 to 2.6 mS/cm. High EC stresses roots through osmotic pressure. Low EC under-feeds plants. Monitor both EC and pH together, as neither reading in isolation gives a complete picture of solution quality.